Hertz experiment
The energy carried by electromagnetic radiation is derived from the kinetic energy of an oscillating charge. To get appreciable amount of radiation, the charge should oscillate to an extent comparable with the wavelength of the radiation. Hertz devised a system in which the charges oscillate with a frequency equal to 5 x 107 Hz, resulting in emission of electromagnetic radiation of wavelength nearly 6 m.
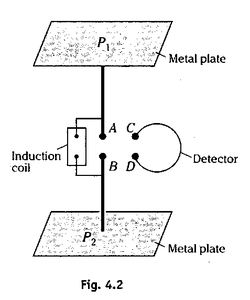
Hertz experimental set up consists of two copper or zinc plates P and P2 placed parallel to each other separated by a distance of 0.6 m (Fig. 4.2). These plates are connected to two well polished metal spheres A and B through thick copper wires. A high potential difference of several thousand volts is applied between the plates by using an induction coil.
Please express your views of this topic A Brief Introduction to Fluid Mechanics by commenting on blog
The air in the small gap between A and B 2 to 3 cm) gets ionized due to high potential difference. This acts as a path for the discharge of the plates. A spark is produced between the spheres A and B. This results in a very high frequency (5 x 107 Hz) oscillations of charges on the plates. Thus a high frequency electric field is produced in the vertical gap between A and B, and a magnetic field of the same frequency is produced in the horizontal plane perpendicular to the gap between the spheres A and B. Electromagnetic waves consisting of these oscillating electric and magnetic fields are radiated from the spark gap. The wavelength of the radiations emitted is nearly 6 m.
Hertz also used a detector to detect the electromagnetic radiations produced in the experiment. Detector consists of an open metallic ring of diameter 0.7 m with small spheres C and D separated by a small gap. The ring is so arranged that it's plane is perpendicular the magnetic field produced by the oscillating charges. The oscillating magnetic field linked with the ring causes a large induced emf which results in a spark between the spheres C and D.
Planck's quantum theory of radiation
Electromagnetic theory failed to explain the photo-electric effect and the energy distribution in the black body spectrum. In 1900 a German Physicist Max Planck putforth a revolutionary theory known as quantum theory of radiation. This theory could successfully explain the energy distribution in the black body spectrum. Later the theory was used by Albert Einstein to explain photo electric effect.
According to quantum theory of radiation emission or absorption of energy does not take place continuously as explained in the classical electromagnetic theory, but in discrete amounts called quanta. Einstein called these quanta as photons when he applied the theory to explain photoelectric emission. The energy of a quantum or photon is given by E = n (hv), where n is an integer, v is the frequency of the radiation to which the photon belongs and '/j' is called Planck's constant whose value is 6.625 x 10~34 Js. That is energy emitted or absorbed by a body is quantised. The least energy of a light wave is hv, where v is the frequency. If the radiation has more energy it must be in terms of integral multiples of hv. Though photons are not material particles, they possess several properties which are same as those of material particles. The photon picture of electromagnetic radiation can be summarized as follows :
1) In interaction with matter, radiation behaves as particles called photons.
2) The energy of each photon of frequency v is E - hv.
3) The photons travel with a speed equal to 'c' the speed of light in free space relative to all frames of reference.
4) Though theoretically the rest mass of a photon is zero, in practice, it can not be zero, since in no frame of reference a photon can be at rest. 2 2 frv
5) The moving mass m of a photon is given by E = mc i.e., hv = mc or m = c
6) Momentum of a photon is, P = mc = — = —, where X is the wavelength of the c X electromagnetic wave.
7) In a photon-particle interaction, the total energy and total momentum are conserved. But the number of photons may not be conserved.
8) During a photon-particle interaction, a photon may be absorbed or a new photon may be created.
9) Electric and magnetic fields can not deflect photons as they are electrically neutral.
Quantum theory of radiation could satisfactorily explain, the black body spectrum, photoelectric effect, compton effect etc.,
Dual nature of light
To explain the properties of light such as interference, diffraction and polarisation, the wave aspect of light should be considered. To account for the experimental results of photoelectric effect, or processes of emission, absorption and scattering one should
consider light to behave as particles (photons). Therefore it has been concluded that light has dual nature i.e., particle and wave nature or wavicle nature. de-Broglie's hypothesis on matter-waves confirmed the dual nature of light. According to de-Broglie, the wavelength of a wave associated with a particle of mass'm' moving with velocity v is given by, X = h/mv. This equation is referred to as de-Broglie's relation and A, is called de-Broglie's wavelength.